 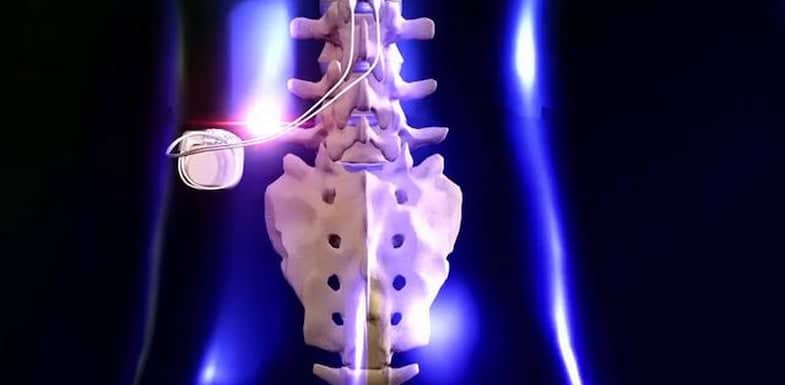 
Some models of this system are Magnetic Resonance (MR) Conditional, and patients with these devices may be scanned safely with magnetic resonance imaging (MRI) when the conditions for safe scanning are met. For more information about MR Conditional neurostimulation components and systems, including equipment settings, scanning procedures, and a complete listing of conditionally approved components, refer to the MRI procedures clinician's manual for neurostimulation systems (available online at medical.abbott/manuals). Advise patients to inform their healthcare professional that they cannot undergo the following procedures:ĭiathermy (short-wave diathermy, microwave diathermy, or therapeutic ultrasound diathermy)Įlectroshock therapy and transcranial magnetic stimulation (TMS) The following procedures are contraindicated for patients with a deep brain stimulation system. This system is contraindicated for patients who meet the following criteria: Unilateral or bilateral stimulation of the ventral intermediate nucleus (VIM) of the thalamus for the suppression of disabling upper extremity tremor in adult essential tremor patients whose tremor is not adequately controlled by medications and where the tremor constitutes a significant functional disability.Bilateral stimulation of the subthalamic nucleus (STN) or the internal globus pallidus (GPi) as an adjunctive therapy to reduce some of the symptoms of advanced levodopa-responsive Parkinson’s disease that are not adequately controlled by medications.The Abbott Medical deep brain stimulation system is indicated for the following conditions: The system is intended to be used with leads and associated extensions that are compatible with the system. This neurostimulation system is designed to deliver low-intensity electrical impulses to nerve structures. 
Deep Brain Stimulation (DBS) Therapy Intended Use 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
